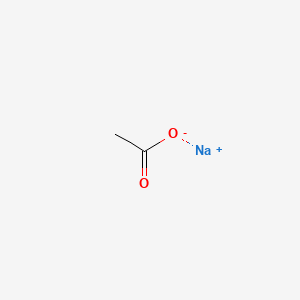
Sodium Acetate (CAS 127-09-3) – Research Grade
Sodium acetate is an organic sodium salt. It contains an acetate.
Research Context
Sodium acetate, a versatile chemical compound, has garnered attention in various research applications due to its unique properties. Its utility spans across fields such as biochemistry, food science, and materials engineering. In biochemistry, sodium acetate serves as a buffering agent, maintaining pH levels in biological experiments. In food science, it acts as a preservative and flavoring agent, enhancing food safety and quality. Additionally, sodium acetate is employed in the production of textiles and as a de-icing agent in winter maintenance.
Mechanism of Action
The mechanism of action of sodium acetate primarily revolves around its role as a buffer. In biological systems, it dissociates into sodium ions and acetate ions, which can interact with hydrogen ions to stabilize pH levels. This property is particularly beneficial in enzymatic reactions where pH fluctuations can adversely affect reaction rates and outcomes. Furthermore, acetate ions can serve as a substrate for various metabolic pathways, influencing energy production and biosynthesis in living organisms.
Solubility and Storage Advice
Sodium acetate is highly soluble in water, making it easy to incorporate into various solutions for experimental purposes. However, it is important to store sodium acetate in a cool, dry place, away from moisture and incompatible substances. Sealed containers made of glass or high-density polyethylene are recommended to prevent contamination and ensure longevity. When handling sodium acetate, standard laboratory safety protocols should be followed to minimize exposure.
Future Research Directions
Future research on sodium acetate is poised to expand its applications, particularly in sustainable practices. Investigating its role in bioenergy, as a potential renewable resource, holds promise for the development of eco-friendly technologies. Additionally, researchers are exploring sodium acetate's efficacy in drug delivery systems, where its properties may enhance the bioavailability of therapeutic agents. With ongoing studies, sodium acetate continues to emerge as a critical component in diverse scientific domains, highlighting its significance and versatility.
Need this compound? Check current availability and pricing.
| CAS Number | 127-09-3 |
|---|---|
| Formula | C2H3NaO2 |
| Mol. Weight | 82.03 g/mol |
| IUPAC Name | sodium acetate |
| Grade | Research Grade |
Technical Support & Logistics
Storage & Handling: Sodium Acetate is strictly allocated for laboratory research use (RUO). Shipped in anti-static packaging.