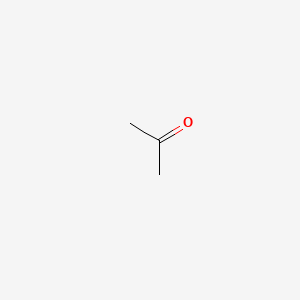
Acetone (CAS 67-64-1) – Research Grade
Acetone is a methyl ketone that consists of propane bearing an oxo group at C2. It has a role as a polar aprotic solvent, a human metabolite and an EC 3.5.1.4 (amidase) inhibitor. It is a methyl ketone, a ketone body, a volatile organic compound and a member of propanones.
Research Context
Acetone, a highly versatile organic solvent, is widely utilized across various fields of chemical research and industrial applications. Its exceptional solvent properties make it an essential reagent in organic synthesis, particularly in the extraction and purification processes. Researchers commonly employ acetone in the formulation of paints, coatings, and adhesives, leveraging its ability to dissolve a wide range of substances.
Research Applications
In laboratory settings, acetone is frequently used to clean glassware and equipment due to its effectiveness in removing organic residues. Furthermore, it plays a critical role in analytical chemistry, serving as a solvent for chromatography techniques, including gas chromatography and high-performance liquid chromatography (HPLC). The compound is also significant in biochemical studies, particularly in the extraction of lipids and other hydrophobic compounds from biological samples.
Mechanism of Action
The mechanism of action of acetone primarily revolves around its polarity and ability to form hydrogen bonds, which facilitates the dissolution of both polar and nonpolar compounds. This property is crucial in various reactions where acetone acts as a solvent, allowing for effective interaction between reactants. Additionally, acetone is known to participate in certain reactions, such as the aldol condensation, where it acts as both a nucleophile and a solvent.
Solubility and Storage Advice
Acetone is miscible with water, ethanol, and many organic solvents, making it a highly effective solvent for a wide array of applications. When storing acetone, it is essential to keep it in a cool, well-ventilated area, away from heat sources and incompatible materials. Containers should be tightly sealed and made of materials compatible with organic solvents, such as glass or specialized plastics.
Future Research Directions
Future research involving acetone is likely to focus on its role in green chemistry initiatives, aiming to reduce environmental impact through the development of sustainable processes that utilize acetone as a solvent. Additionally, exploring its potential applications in nanotechnology and the synthesis of new materials can pave the way for innovative discoveries. Continued investigation into the metabolic pathways involving acetone in biological systems could also yield important insights into its physiological effects and therapeutic potentials.
Need this compound? Check current availability and pricing.
| CAS Number | 67-64-1 |
|---|---|
| Formula | C3H6O |
| Mol. Weight | 58.08 g/mol |
| IUPAC Name | propan-2-one |
| Grade | Research Grade |
Technical Support & Logistics
Storage & Handling: Acetone is strictly allocated for laboratory research use (RUO). Shipped in anti-static packaging.