Uric Acid (CAS 69-93-2) – Research Grade
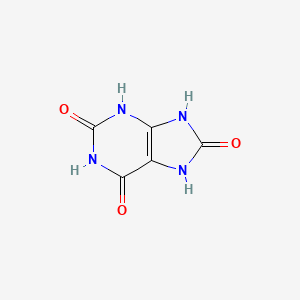
7,9-dihydro-1H-purine-2,6,8(3H)-trione is an oxopurine in which the purine ring is substituted by oxo groups at positions 2, 6, and 8. It has a role as a human metabolite, an Escherichia coli metabolite and a mouse metabolite. It is a tautomer of a 2,6-dihydroxy-7,9-dihydro-8H-purin-8-one, a 9H-purine-2,6,8-triol, a 7H-purine-2,6,8-triol, a 1H-purine-2,6,8-triol and a 5,7-dihydro-1H-purine-2,6,8(9H)-trione.
Research Context
Uric acid, a key product of purine metabolism, plays a significant role in various biological processes and has garnered considerable attention in scientific research. Due to its dual role as an antioxidant and a pro-oxidant, uric acid has been implicated in the pathogenesis of several diseases, including gout, cardiovascular diseases, and metabolic syndrome. Its unique biochemical properties make it a critical subject of study in both clinical and laboratory settings.
Research Applications
Researchers have utilized uric acid in various applications, particularly in understanding its role in oxidative stress and inflammation. Its ability to scavenge free radicals positions it as a potential therapeutic agent for conditions such as neurodegenerative diseases. Moreover, uric acid levels are often monitored as indicators of renal function and metabolic disorders, providing valuable insights into patient health and disease progression.
Mechanism of Action
The mechanism of action of uric acid is complex and multifaceted. It is known to influence endothelial function and modulate inflammatory pathways. Elevated levels of uric acid can lead to the activation of pro-inflammatory cytokines, potentially contributing to the development of chronic diseases. Conversely, its antioxidant properties may protect against cellular damage, suggesting a delicate balance in its physiological roles.
Solubility and Storage Advice
Uric acid exhibits low solubility in water, which can complicate its handling in research applications. To maintain stability, it is recommended to store uric acid in a cool, dry place, away from direct light. Solutions should be prepared fresh when possible, and any diluted samples should be used promptly to avoid degradation.
Future Research Directions
Future research on uric acid is expected to focus on elucidating its role in various pathophysiological conditions, particularly its paradoxical nature as both a harmful and beneficial molecule. Investigations into the therapeutic potential of uric acid modulation, as well as its interactions with other biomolecules, hold promise for developing novel treatment strategies. Additionally, the exploration of uric acid's genetic and environmental influences may unveil new insights into its implications in human health.
Need this compound? Check current availability and pricing.
| CAS Number | 69-93-2 |
|---|---|
| Formula | C5H4N4O3 |
| Mol. Weight | 168.11 g/mol |
| IUPAC Name | 7,9-dihydro-3H-purine-2,6,8-trione |
| Grade | Research Grade |
Technical Support & Logistics
Storage & Handling: Uric Acid is strictly allocated for laboratory research use (RUO). Shipped in anti-static packaging.